Directeur de Recherche

Laboratoire LSAMM
Université de Strasbourg
Institut Le Bel
4, rue Blaise Pascal
67000 Strasbourg-France
email: Henri-Pierre J. de Rouville
-
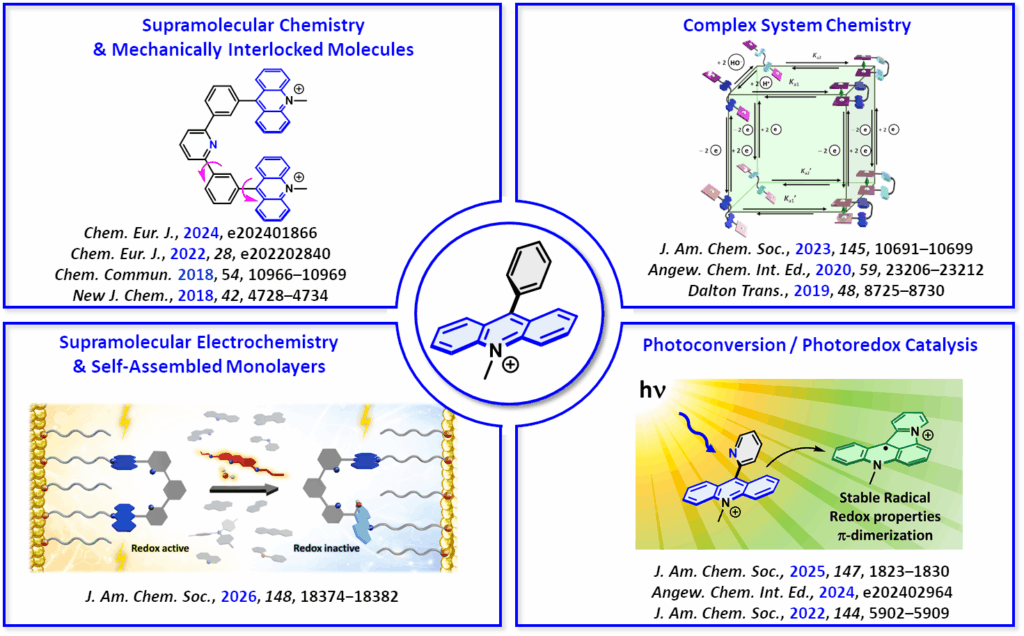
Research Interests
-
Professional Experience
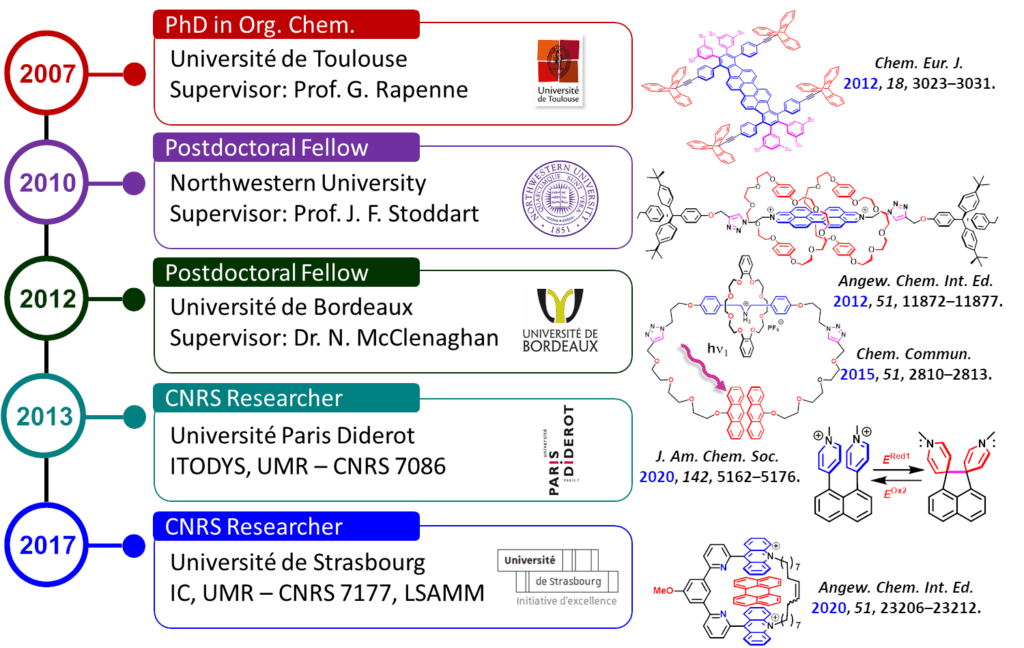
Henri-Pierre Jacquot de Rouville graduated from the University Paul Sabatier of Toulouse. In 2007, he joined C. Joachim’s Group and obtained a PhD degree for his synthesis of technomimetic molecules for nanomechanical applications under the supervision of Professor G. Rapenne (CEMES-CNRS, Toulouse). From 2010 to 2012, he joined Professor J. F. Stoddart’s Group as a postdoctoral fellow where he investigated the chemistry of mechanically interlocked molecules. Then, he moved back to France where he worked as a postdoctoral fellow with N. McClenaghan (ISM, Bordeaux). In 2013, he was appointed as Chargé de Recherche (Full Time Researcher at the CNRS) at the laboratory ITODYS, Paris. In November 2017, he moved at the Institut de Chemistry of Strasbourg where he joined the LSAMM research group.
-
Education
2022: Habilitation à Diriger des Recherches
Université de Strasbourg, Institut de Chimie, UMR – CNRS 7177 (Strasbourg, France)
« Positively Charged Aromatic Systems: Electrochemical and Recognition Properties »
2007 – 2010: PhD in Organic Chemistry
Université Paul Sabatier, CEMES, UPR – CNRS 8011 (Toulouse, France)
Supervisor: Prof. Gwénaël Rapenne
« Design of Technomimetic Molecules for Applications in Nanomechanics »
2007: MSc in Molecular Chemistry with high honors (1st/43)
Université Paul Sabatier (Toulouse, France)
2005: BSc in Molecular Chemistry
Université Paul Sabatier (Toulouse, France)
-
Publications
42. Tandem Guest-Host Switching for Chemical-to-Electrochemical Signal Transduction
L. Lefrançois, E. S. Gauthier, S. Chapuis, X. Shen, M. Ely Namoro, C. Gourlaouen, A. Chaumont, V. Heitz, L. Ruhlmann,* M. Frasconi,* M. Hromadová,* H.-P. Jacquot de Rouville* J. Am. Chem. Soc., 2026, 148, 18374−18382. (Link)
41. Path Selective Photoinduced Energy and Electron Transfer in a Bis(acridinium-Zn(II) Porphyrin)-tetrapyridyl Porphyrin Host-Guest Complex (open access)
F. Ruani, D. Veclani, D. Sanchez-Resa, A. Edo-Osagie, G. Creste, A. Barbieri, V. Heitz,* H.-P. Jacquot de Rouville,* N. Armaroli,* B. Ventura* Chem. Eur. J., 2025, e02604. (Link)
40. Porphyrin and triphenylamine cores linked by a phenyl-azomethine bridge: synthesis and characterization
I. P. Mangaa, M. Lo,* A. F. Fall Pouyea, C. Gourlaouen, H.-P. Jacquot de Rouville, A. Gassama, J. Weiss Journal of Porphyrins and Phthalocyanines, 2025, doi: 10.1142/S1088424625500932. (Link)
39. Long-Chain Per-/Poly-Fluoroalkyl Substances (PFASs) and Their Supramolecular Interactions (invited)
L. Lefrançois, V. Heitz, J.-M. Vincent, H.-P. Jacquot de Rouville* Trends in Chemistry, 2025, 7, 106–109. (Link)
38. Viridium: A Stable Radical and its π-Dimerization
H.-P. Jacquot de Rouville,* C. Gourlaouen,* D. Bardelang, N. Le Breton, J. S. Ward, L. Ruhlmann, J.-M. Vincent, D. Jardel, K. Rissanen, J.-L. Clément, S. Choua, V. Heitz J. Am. Chem. Soc., 2025, 147, 1823–1830. (Link)
37. π-mers and π-dimers: Two Radical Supramolecular Interactions – A Tutorial Review
J. Joseph, M. Berville, J. Wytko, J. Weiss, H.-P. Jacquot de Rouville* Chem. Eur. J., 2024, e202403115. (Link)
36. Self-Assembled Bis-Acridinium Tweezer Equilibria Controlled by Multi-Responsive Properties
J. Hu, J.-P. Launay, A. Chaumont, V. Heitz,* H.-P. Jacquot de Rouville* Chem. Eur. J., 2024, e202401866. (Link)
35. Photogeneration of Chlorine Radical from a Self-Assembled Fluorous 4CzIPN•Chloride Complex: Application in C–H Bond Functionalization
V. Carré, P. Godard, R. Méreau, H.-P. Jacquot de Rouville, G. Jonusauskas, N. McClenaghan, T. Tassaing, J.-M. Vincent* Angew. Chem. Int. Ed., 2024, e202402964. (Link)
34. Mixed thioether/amine-functionalized expanded calixarenes, related macropolycycles, and metal complexes thereof
J. Taut, F. Schleife, M. Börner, C. Bonnot, H.-P. Jacquot de Rouville, J.-C. Chambron,* B. Kersting* Eur. J. Inorg. Chem., 2024, e202300580. (Link)
33. A Multi-Responsive 8-State bis(Acridinium-Zn(II) Porphyrin) Receptor
A. Edo-Osagie, D. Serillon, F. Ruani, X. Barril, C. Gourlaouen, N. Armaroli,* B. Ventura,* H.-P. Jacquot de Rouville,* V. Heitz* J. Am. Chem. Soc., 2023, 145, 10691–10699. (Link)
32. Crystal Engineering, Electron Conduction, and Molecular Recognition and Reactivity by Chalcogen Bonds in Tetracyanoquinodimethanes Fused with [1,2,5]Chalcogenadiazoles
T. Shimajiri,* H.-P. Jacquot de Rouville, V. Heitz, Tomoyuki Aktagawa, Takanori Fukushima, Yusuke Ishigaki, T. Suzuki* Synlett, 2023, DOI: 10.1055/a-2072-2951. (Link)
31. Photophysical characterization of a bisacridinium-diphenylporphyrin conjugate
F. Ruani, A. Edo-Osagie, H.-P. Jacquot de Rouville, V. Heitz, B. Ventura, N. Armaroli Journal of Porphyrins and Phthalocyanines, 2023, doi: 10.1142/S1088424623500396. (Link)
30. Dual-Readout of the Mechanical Response of a bis-Acridinium [2]Rotaxane
J. Hu, S. Adrouche, E. S. Gauthier, N. Le Breton, M. Cecchini, C. Gourlaouen, S. Choua, V. Heitz,* H.-P. Jacquot de Rouville* Chem. Eur. J., 2022, 28, e202202840. (Link) Front Cover
29. Solid-State Assembly by Chelating Chalcogen Bonding in Quinodimethane Tetraesters Fused with a Chalcogenadiazole
Y. Ishigaki, K. Asai, H.-P. Jacquot de Rouville, T. Shimajiri, J. Hu, V. Heitz, T. Suzuki* ChemPlusChem, 2022, e202200075. (Link)
28. Photoinduced Arylation of Acridinium Salts: Tunable Photoredox Catalysts for C−O Bond Cleavage
Y-X. Cao, G. Zhu, Y. Li, N. Le Breton, C. Gourlaouen, S. Choua, J. Boixel,* H.-P. Jacquot de Rouville,* J.-F. Soulé* J. Am. Chem. Soc., 2022, 144, 5902–5909. (Link)
27. On the Supra-LUMO Interaction: Case Study of a Sudden Change of Electronic Structure as a Functional Emergence
A. Gosset, Š. Nováková Lachmanová, S. Cherraben, G. Bertho, J. Forté, C. Perruchot, H.-P. Jacquot de Rouville, L. Pospíšil, M. Hromadová,* É. Brémond,* P. P. Lainé,* Chem. Eur. J., 2021, 27, 17889–17899 (Link).
26. Synthesis, electronic and photophysical properties of a bisacridinium-Zn(II) porphyrin conjugate
A. Edo-Osagie, D. Sánchez-Resa, D. Serillon, E. Bandini, C. Gourlaouen, H.-P. Jacquot de Rouville,* B. Ventura,* V. Heitz,* C. R. Chimie, 2021, 24, 1–9 (Link).
25. Molecular Recognition by Chalcogen Bond: Selective Charge-transfer Crystal Formation of Dimethylnaphthalene with Selenadiazolotetracyano naphthoquinodimethane
Y. Ishigaki, K. Asai, H.-P. Jacquot de Rouville, T. Shimajiri, V. Heitz, H. Fujii-Shinomiya, T. Suzuki,* Eur. J. Org. Chem., 2021, 6, 990–997 (Link).
24. N-substituted Acridinium as a Multi-Responsive Recognition Unit in Supramolecular Chemistry
H.-P. Jacquot de Rouville,* J. Hu, V. Heitz,* ChemPlusChem, 2021, 86, 110–129 (Link).
23. A Bis-Acridinium Macrocycle as Multi-Responsive Receptor and Selective Phase Transfer Agent of Perylene
J. Hu, J. S. Ward, A. Chaumont, K. Rissanen, J.-M. Vincent,* V. Heitz,* H.-P. Jacquot de Rouville,* Angew. Chem. Int. Ed., 2020, 59, 23206–23212 (Link).
22. Electron Storage System Based on a Two-Way Inversion of Redox Potentials
A. Gosset, L. Wilbraham, Š. Nováková Lachmanová, R. Sokolová, G. Dupeyre, F. Tuyèras, P. Ochsenbein, C. Perruchot, H.-P. Jacquot de Rouville, H. Randriamahazaka,* L. Pospíšil,* I. Ciofini,* M. Hromadová,* Philippe P. Lainé,* J. Am. Chem. Soc., 2020, 142, 5162–5176 (Link).
21. Self-Complementary and Narcissistic Self-Sorting of bis-Acridinium Tweezers
H.-P. Jacquot de Rouville,* C. Gourlaouen, V. Heitz,* Dalton Trans., 2019, 48, 8725–8730 (Link).
20. Surface manipulation of a curved polycyclic aromatic hydrocarbon-based nanovehicle molecule equipped with triptycene wheels
W.-H. Soe,* C. Duranda, O. Guillermet, S. Gauthier, H.-P. Jacquot de Rouville, S. Srivastava, C. Kammerer, G. Rapenne, C. Joachim,* Nanotechnology, 2018, 29, 495401 (Link).
19. Entwined Dimers Formation from Self-Complementary bis-Acridiniums
H.-P. Jacquot de Rouville,* N. Zorn, E. Leize-Wagner, V. Heitz,* Chem. Commun., 2018, 54, 10966–10969 (Link).
18. From the Synthesis of Nanovehicles to Participation in the First Nanocar Race—View from the French Team
H.-P. Jacquot de Rouville, C. Kammerer, G. Rapenne,* Molecules, 2018, 23(3), 612 (Link).
17. A Chemically-Responsive bis-Acridinium Receptor
A. Gosset, Z. Xu, F. Maurel,* L.-M. Chamoreau, S. Nowak, G. Vives, C. Perruchot, V. Heitz,* H.-P. Jacquot de Rouville,* New J. Chem., 2018, 42, 4728–4734 (Link).
16. Photodriven [2]Rotaxane-[2]Catenane Interconversion
A. Tron, H.-P. Jacquot de Rouville, A. Ducrot, J. H. R. Tucker,* M. Baroncini, A. Credi,* N. D. McClenaghan,* Chem. Commun., 2015, 51, 2810–2813. (Link)
15. Formation of a Hydrogen-bonded Barbiturate [2]-Rotaxane
A. Tron, P. Thornton, M. Rocher, H.-P. Jacquot de Rouville, J.-P. Desvergne, B. Kauffmann, T. Buffeteau, D. Cavagnat, J. Tucker,* N. McClenaghan,* Org. Lett., 2014, 16, 1358–1361. (Link)
14. An Electrochemically and Thermally Switchable Donor-Acceptor [c2]Daisy Chain Rotaxane
C. J. Bruns, J. Li, M. Frasconi, S. T. Schneebeli, J. Iehl, H.-P. Jacquot de Rouville, S. I. Stupp, G. A. Voth,* J. F. Stoddart,* Angew. Chem. Int. Ed., 2014, 53, 1953–1958. (Link)
13. π–Dimerization of viologen subunits around the core of C60 from twelve to six directions
J. Iehl, M. Frasconi, H.-P. Jacquot de Rouville, N. Renaud, S. M. Dyar, N. L. Strutt, R. Carmieli, M. R. Wasielewski, M. A. Ratner, J.-F. Nierengarten, J. F. Stoddart,* Chem. Sci., 2013, 4, 1462–1469. (Link)
12. Chameleonic Binding of the Dimethyldiazaperopyrenium Dication by Cucurbit[8]uril
K. J. Hartlieb, A. N. Basuray, C. Ke, A. A. Sarjeant, H.-P. Jacquot de Rouville, T. Kikuchi, R. S. Forgan, J. W. Kurutz, J. F. Stoddart,* Asian J. Org. Chem., 2013, 2, 225–229. (Link)
11. Beyond Perylene Diimides – Diazaperopyrenium as Chameleonic Nanoscale Building Blocks
A. N. Basuray, H.-P. Jacquot de Rouville, K. J. Hartlieb, A. C. Fahrenbach, J. F. Stoddart,* Chem. Asian. J., 2013, 8, 524–532. (Link)
10. The Chameleonic Nature of Diazaperopyrenium Recognition Processes
A. N. Basuray, H.-P. Jacquot de Rouville, K. J. Hartlieb, T. Kikuchi, N. L. Strutt, C. J. Bruns, M. W. Ambrogio, A.-J. Avestro, S. T. Schneebeli, A. C. Fahrenbach, J. F. Stoddart,* Angew. Chem. Int. Ed., 2012, 51, 11872–11877. (Link)
9. A Naphthalene Diimide [2]Rotaxane
H.-P. Jacquot de Rouville, J. Iehl, C. J. Bruns, P. L. McGrier, M. Frasconi, A. A. Sarjeant, J. F. Stoddart, Org. Lett., 2012, 14, 5188–5191. (Link)
8. STM Manipulation of Boron-Subphthalocyanine Nano-Wheel Dimers on Au(111)
A. Nickel, J. Meyer, R. Ohmann, H.-P. Jacquot de Rouville, G. Rapenne, C. Joachim, G. Cuniberti, F. Moresco,* J. Phys.: Condens. Matter., 2012, 24, 404001. (Link)
7. Synthesis and STM Imaging of Symmetric and Dissymmetric Ethynyl-Bridged Dimers of Boron-Subphthalocyanine Bowl-Shaped Nano-Wheels
H.-P. Jacquot de Rouville, R. Garbage, F. Ample, A. Wadewitz, J. Meyer, F. Moresco, C. Joachim, G. Rapenne,* Chem. Eur. J., 2012, 18, 8925–8928. (Link)
6. Synthesis of Polycyclic Aromatic Hydrocarbon-Based Nanovehicles Equipped with Triptycene Wheels
H.-P. Jacquot de Rouville, R. Garbage, R. E. Cook, A. R. Pujol, A. M. Sirven, G. Rapenne,* Chem. Eur. J., 2012, 18, 3023–3031. (Link)
5. A Neutral Redox-Switchable [2]Rotaxane
J.-C. Olsen, A. C. Fahrenbach, A. Trabolsi, D. C. Friedman, S. K. Dey, C. M. Gothard, A. K. Shveyd, T. B. Gasa, J. M. Spruell, M. A. Olson, C. Wang, H.-P. Jacquot de Rouville, Y. Y. Botros, J. F. Stoddart,* Org. Biomol. Chem., 2011, 9, 7126–7133. (Link)
4. Synthesis of a Photoswitchable Azobenzene-Functionalized Tris(indazol-1-yl)borate Ligand and its Ruthenium(II) Cyclopentadienide Complex
H.-P. Jacquot de Rouville, D. Villenave, G. Rapenne,* Tetrahedron, 2010, 66, 1885–1891. (Link)
3. Prototypes of Molecular Motors Based on Star-Shaped Organometallic Ruthenium Complexes
G. Vives, H.-P. Jacquot de Rouville, A. Carella, J.-P. Launay, G. Rapenne,* Chem. Soc. Rev., 2009, 6, 1551–1561. (Link)
2. Synthesis and Analytical Resolution of Chiral Pyrazoles from Dihydrocarvone
H.-P. Jacquot de Rouville, G. Vives, E. Tur, J. Crassous, G. Rapenne,* New J. Chem., 2009, 2, 293–299. (Link)
1. A Family of Electron-Triggered Molecular Motors Based on Aromatic Building Blocks
H.-P. Jacquot de Rouville, G. Vives, G. Rapenne,* Pure Appl. Chem., 2008, 80, 659–667. (Link)